 Gradient gels are another great option to enhance resolution. Under these conditions, all of the proteins in a sample migrate through the stacking gel at the same pace and enter the resolving gel at the same time where they are then separated by size. 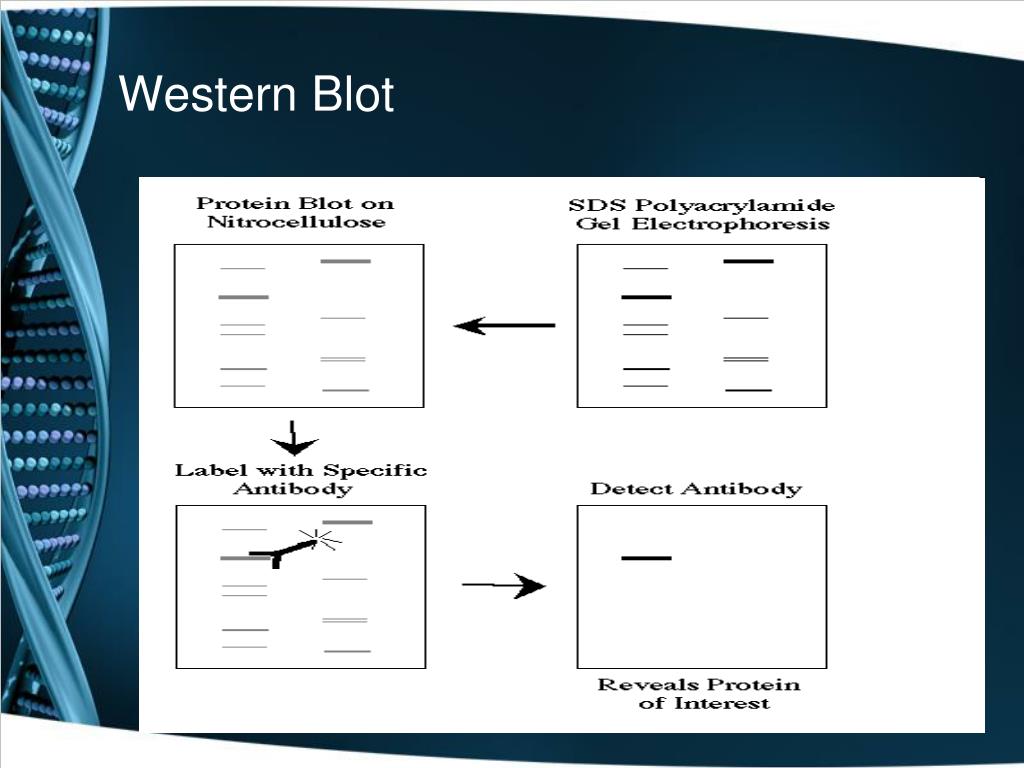
A stacking gel typically has a different ionic strength and lower pH and acrylamide content than the resolving gel. To further improve resolution, use a stacking gel on top of the resolving gel. For large proteins, reduce the percentage of acrylamide. For small proteins, use a higher percentage of acrylamide to increase migration resistance and improve separation. 
For better protein separation, vary the acrylamide content of the gel. Smaller proteins encounter less resistance in the acrylamide matrix and migrate faster through the gel. An electric current applied to the gel causes the negatively charged proteins to migrate toward the positive charge at the bottom of the gel and separate by size. In addition to the samples, a protein standard of known molecular weight in a separate lane can help confirm the size of the protein of interest. Load the samples, one sample per lane, onto the top of a resolving gel composed of the crosslinked polymer acrylamide. Denature the samples to their primary amino acid sequence by boiling in the presence of a reducing agent, typically containing thiols, to cleave disulfide bonds. To prepare the samples for SDS-PAGE, measure the protein content and normalize to ensure equivalent loading. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) is the first step of a western. For the purpose of this blog, we will be focusing on denaturing westerns. In a denaturing western, the protein is denatured to its primary structure and separated by size with smaller molecules moving more quickly through the matrix. In a native western, the protein’s secondary and tertiary structures remain intact and the protein is separated through a matrix by charge. 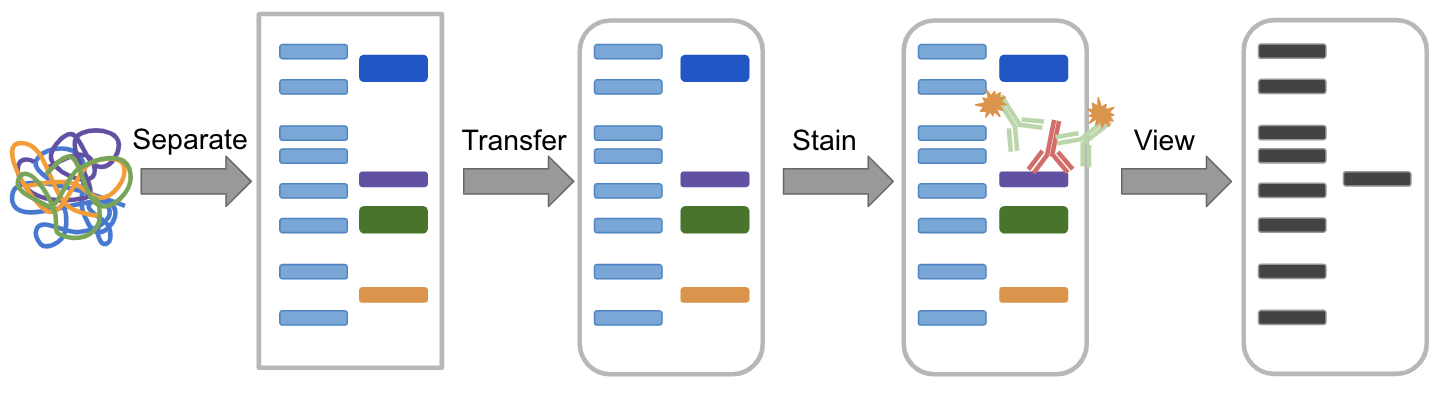
Westerns are divided into two categories, native and denaturing. Westerns are useful not only to detect the presence or absence of a protein, but can also determine if proteins are being up- or downregulated in a system, detect post-translational modifications, quantify protein levels relative to standards, detect the cellular location of proteins, and can be a readout for protein interaction studies such as immunoprecipitation and pull-down assays. This process allows you to detect a single, specific protein within the complex mix derived from cells or tissues. Starting with a mixture of proteins, it is separated in a gel, transferred to a membrane, stained with antibodies, and visualized. Figure 1: Overview of the Western blot process. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
